Location: Home >> Detail
TOTAL VIEWS
Department of Medicine and Oncology, Division of Geriatric Medicine, San Luigi Gonzaga Hospital, 10043 Orbassano, Turin, Italy
Introduction: Demographic aging has led to an increase in “oldest-old” patients (over-85) characterized by high frailty and multimorbidity. In this scenario, the use of drugs with anticholinergic properties for managing behavioral and psychological symptoms of dementia (BPSD) and chronic comorbidities generates a critical anticholinergic burden, a potential determinant of adverse outcomes such as delirium and functional decline. This study aims to analyze the correlations between anticholinergic burden, sarcopenia, and delirium in a cohort of acute geriatric patients. Materials and Methods: A retrospective observational study was conducted on 60 patients admitted urgently to a Geriatrics unit for infectious or inflammatory events. At baseline, a Comprehensive Geriatric Assessment (CGA) was performed, including: Charlson Comorbidity Index, Barthel Index and IADL (functional status), MMSE (cognition), 4AT (delirium screening), and ultrasonographic evaluation of the quadriceps muscle (sarcopenia). The anticholinergic burden (ACB scale) was calculated at admission and discharge to evaluate the impact of geriatric intervention and medication reconciliation. Results: The study population (mean age 87.9 ± 5.5 years) was characterized by severe frailty (mean CFS: 6.8; mean Charlson Comorbidity Index: 10.3). Specialist geriatric intervention was associated with a significant reduction in the mean anticholinergic burden (ACB), which decreased from 4.23 ± 1.5 at admission to 2.78 ± 1.2 at discharge (p < 0.0001; Cohen’s d = 1.05, indicating a large clinical effect). At admission, a high ACB score was significantly associated with the occurrence of delirium in patients with sepsis. Specifically, the anticholinergic burden showed a strong inverse correlation with cognitive performance (MMSE: r = −0.51; 95% CI [−0.68, −0.29]; p < 0.001) and a significant positive association with ultrasound-detected sarcopenia (r = 0.49; 95% CI [0.27, 0.66]; p < 0.001). Furthermore, exposure to high-risk anticholinergic drugs appeared to be linked to higher delirium severity scores (4AT: r = 0.42; 95% CI [0.19, 0.61]; p = 0.0015). Conclusions: This study suggests that the care burden in septic elderly patients is influenced by multidimensional factors. Sarcopenia and pre-existing cognitive impairment appear to represent substrates of vulnerability upon which anticholinergic burden may act as a contributory factor in the development of delirium. Systematic deprescribing and medication reconciliation emerge as priority clinical and ethical strategies to improve clinical outcomes and promote deinstitutionalization.
The current demographic landscape, marked by a progressive and inexorable demographic ageing, has resulted in an exponential increase in hospital admissions involving the oldest-old (over-85) [1]. This specific cohort of patients characteristically presents in a state of severe frailty in which even a minor stressful event can trigger cascades of irreversible functional and cognitive decline, as the physiological reserve is no longer capable of compensating for the resulting disequilibrium [2].
In these individuals, who are often multimorbid, acute infections such as pneumonia, urinary tract infections, or septic states do not represent simple clinical challenges; rather, they act as catalysts that expose biological reserves already compromised by chronic comorbidities and the resulting polypharmacy [3].
Concurrently, increased life expectancy has led to a rise in behavioral disorders and dementias, a trend that epidemiological projections indicate will grow substantially starting in 2025 [4]. This healthcare emergency has complicated therapeutic management, contributing to widespread use of antipsychotics, antidepressants, and neuroleptics to manage the non-cognitive symptoms of dementia (BPSD) [5]. However, a notable clinical paradox emerges: the very drugs required for behavioural control possess marked anticholinergic properties. These anticholinergic effects compound those of medications prescribed for common comorbidities such as hypertension or urinary incontinence, collectively contributing to what is termed the “anticholinergic burden” [6,7].
The weight of this cumulative load is such that the World Health Organization (WHO) has issued numerous alerts regarding polypharmacy in the elderly, formally urging the medical community toward the systematic deprescribing of high-risk medications [8].Particular attention is focused on antipsychotics and antidepressants with high anticholinergic activity, as their cumulative effect is a predictor of adverse outcomes ranging from falls and fractures to aspiration pneumonia and a significant increase in mortality risk [9,10]. Among the most serious manifestations of this pharmacological excess is delirium, an acute neuropsychiatric syndrome that can precipitate a rapid and potentially irreversible deterioration in cognitive function [7,11]. Although the etiology of delirium is multifactorial [12,13], anticholinergic burden is currently considered the primary modifiable precipitant amenable to clinical intervention [14].
In this context, tools such as the Anticholinergic Cognitive Burden (ACB) scale allow for the quantification of risk and the prediction of global functional impairment, making medication review an ethical and clinical necessity. Based on these premises, the objective of this study is to analyze—within a population of frail oldest-old patients—the correlations between frailty (evaluated in its two key domains: cognitive, as expressed through delirium, and physical, as expressed through sarcopenia) and the pharmacological anticholinergic burden at the time of admission to an acute geriatrics unit due to an infectious or inflammatory pathology requiring hospitalization.
This retrospective observational study was conducted at the Geriatrics Operating Unit of the San Luigi Gonzaga University Hospital. The study population consisted of 60 patients aged 65 years or older, consecutively enrolled between January and November 2025. All subjects were urgently admitted via the hospital’s Emergency Department with a confirmed diagnosis of an acute infectious or septic event. A consecutive sampling strategy was adopted to minimize selection bias and ensure that the cohort accurately reflected the clinical reality of acutely ill geriatric patients.
Regarding data integrity, a complete-case analysis approach was followed; patients with missing or incomplete clinical data were excluded from the final sample. Notably, the incidence of missing data was minimal, affecting less than 2% of the initial population.
Clinical Characteristics and Setting of OriginThe diagnostic landscape of the cohort was predominantly characterized by respiratory tract involvement, accounting for 85% (n = 51) of cases. The distribution included pneumonia (n = 24, 40.0%), acute respiratory infections (n = 13, 21.7%), exacerbated Chronic Obstructive Pulmonary Disease (COPD) (n = 9, 15.0%), multi-focal pneumonia (n = 3, 5.0%), and acute respiratory failure due to COVID-19 (n = 2, 3.3%). Extrapulmonary infections comprised a minority of the sample, including sepsis of unknown origin (n = 5, 8.3%), soft tissue infections (n = 2, 3.3%), abdominal sepsis (n = 1, 1.7%), and osteomyelitis (n = 1, 1.7%).
Regarding the clinical setting of origin, 55% (n = 33) of patients were admitted from their private domicile without pre-existing formal home care. The remaining portion exhibited a high level of prior frailty, with 33.3% (n = 20) transferred from Residential Care Facilities (RSA) and 11.7% (n = 7) under Integrated Home Care (ADI) programs.
Comprehensive Geriatric Assessment (CGA)To ensure methodological rigor, every patient underwent a standardized Comprehensive Geriatric Assessment (CGA) at the time of admission to establish a multidimensional baseline. General clinical status was quantified using the Charlson Comorbidity Index, while functional status was mapped via the Barthel Index (ADL) and the IADL index.
The neuropsychological sphere was evaluated by integrating the Mini-Mental State Examination (MMSE) for baseline cognitive status with the 4AT standardized protocol for timely delirium screening. This integrated approach was essential for detecting acute alterations in attention and alertness typical of the hospitalized elderly.
Sarcopenia Assessment ProtocolPhysical frailty was investigated through an ultrasound-based sarcopenia assessment performed at the bedside. To minimize inter-observer variability, all measurements were performed by a single, highly experienced operator (T.F.), a geriatrician with expertise in Point-of-Care Ultrasound (POCUS). Assessments were conducted using an Esaote MyLab 90 ultrasound system equipped with a 7.5 MHz linear probe.
Patients were examined in a supine position with the lower limbs slightly flexed to ensure relaxation of the quadriceps musculature. The high-frequency linear transducer was set to a musculoskeletal/deep muscle preset. Following validated methodological recommendations, the operator positioned the probe at the knee level and adjusted it to obtain optimal longitudinal and transverse images of the vastus lateralis thickness and pennation angle.
Pharmacological Reconciliation and Anticholinergic BurdenThe core of the pharmacological analysis involved a dynamic evaluation of the Anticholinergic Burden (ACB), treated as the independent variable. The medication reconciliation protocol was performed by a single specialist (T.F) in two distinct phases:
1.
2.
High Anticholinergic Burden was specifically defined by an ACB score > 3 at admission, a threshold recognized as a critical predictor of cognitive impairment and falls. Prescription optimization and deprescribing strategies were strictly guided by the AGS Beers Criteria®, targeting high-risk classes such as first-generation antihistamines, benzodiazepines, tricyclic antidepressants, and proton pump inhibitors (PPIs). ACB scores were calculated at both time points to facilitate a longitudinal comparison. These results were subsequently analyzed in relation to dependent variables, including the incidence of delirium, length of hospital stay (LOS), and post-discharge management complexity.
Risk Stratification: The High-Risk GroupTo further stratify clinical vulnerability, a “High-Risk Group” was defined based on a synergistic model of physical and pharmacological frailty. This group included patients simultaneously presenting with Severe Sarcopenia (ultrasound quadriceps thickness < 10.5 mm, representing the bottom 25th percentile) and a High Anticholinergic Burden (ACB > 3). This combined threshold identified the subset of patients at the highest risk for adverse outcomes.
Statistical AnalysisStatistical analysis was performed using R Statistical Software (version 4.3.1). The normality of continuous variables was assessed using the Shapiro-Wilk test. Parametric tests (Student’s t-test) were employed for normally distributed data, while non-parametric alternatives (Mann-Whitney U and Wilcoxon signed-rank tests) were used when the normality assumption was violated.
To complement p-values, 95% Confidence Intervals (95% CI) were calculated for all correlation coefficients and mean differences. Effect sizes were estimated to quantify the clinical magnitude of findings: Cohen’s d for group comparisons and the coefficient of determination (r2) for correlations. For non-parametric group comparisons, the effect size r was calculated as Z/√n. Statistical significance was set at p < 0.05.
Statistical AnalysisStatistical analysis was performed using R Statistical Software (version 4.3.1; R Foundation for Statistical Computing, Vienna, Austria). The normality of continuous variables was formally assessed using the Shapiro-Wilk test, which is particularly sensitive for small to moderate sample sizes (n < 100).
For normally distributed data, we employed parametric tests (Student’s t-test for independent or paired samples, as appropriate). When the normality assumption was violated, non-parametric alternatives were used: the Mann-Whitney U test for comparisons between independent groups and the Wilcoxon signed-rank test for longitudinal analysis of the anticholinergic burden (admission vs. discharge).
To complement p-values, 95% Confidence Intervals (95% CI) were calculated for all correlation coefficients and mean differences. To quantify the clinical magnitude of our findings beyond statistical significance, effect sizes were estimated: Cohen’s d was used for group comparisons (interpreted as 0.2 = small, 0.5 = medium, and 0.8 = large), while the coefficient of determination (r2) was reported for correlations to quantify the proportion of variance shared between the anticholinergic burden and clinical outcomes (cognitive and physical scores). For non-parametric group comparisons, the effect size r was calculated as Z/√n. Statistical significance was set at p < 0.05. To identify the independent association between anticholinergic load and delirium, a multivariable logistic regression model was constructed. Delirium incidence was treated as the dependent variable, while the ACB score at admission was entered as the primary independent predictor, adjusting for age and the Charlson Comorbidity Index as potential clinical confounders.
Data were analyzed using non-parametric tests (Wilcoxon) for pre-post mean comparisons and Pearson/Spearman correlation coefficients to identify links between clinical variables and the Burden.
The analyzed population had a mean age of 87.9 years (±5.5), with a mean hospital stay of 21.8 days. The clinical profile highlighted severe frailty, with a mean Clinical Frailty Scale (CFS) score of 6.8 and a Charlson Index of 10.3, indicating a high number of concomitant pathologies (See Table 1).
Evolution of Care Burden
A significant clinical-care improvement was observed during hospitalization. The Anticholinergic Burden score dropped from a mean of 4.23 at admission to 2.78 at discharge (p < 0.0001). Despite this, 60% of patients experienced episodes of delirium, a complication associated with prolonged stay and a lower probability of immediate return to home (which occurred in 55% of cases). (Table 2; Figures 1 and 2).
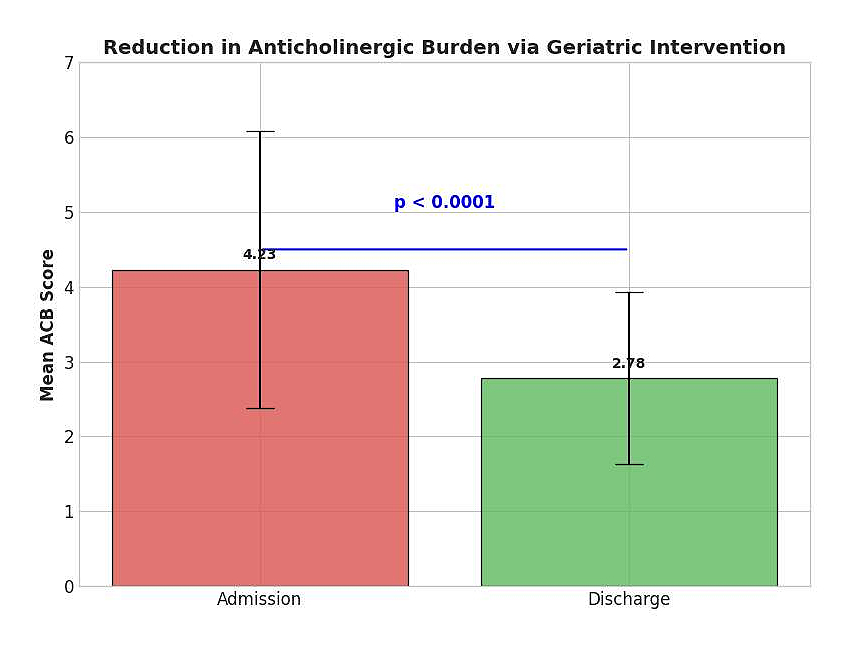 Figure 2. The “Vulnerability-Trigger” Longitudinal reduction of mean ACB score from admission to discharge. This histogram illustrates the significant reduction in the mean ACB score from admission to discharge. This change (4.23 ± 1.85 vs. 2.78 ± 1.15) reflects the success of the medication reconciliation and deprescribing strategies implemented during the hospital stay.
Figure 2. The “Vulnerability-Trigger” Longitudinal reduction of mean ACB score from admission to discharge. This histogram illustrates the significant reduction in the mean ACB score from admission to discharge. This change (4.23 ± 1.85 vs. 2.78 ± 1.15) reflects the success of the medication reconciliation and deprescribing strategies implemented during the hospital stay.
Determinants of Burden and Anticholinergic Role
Correlation analysis identified the following drivers of care burden at admission:
●
●
●
To control for potential confounders, a logistic regression model was performed with delirium as the dependent variable. After adjusting for age and Charlson Comorbidity Index, the Anticholinergic Burden remained a significant factor associated with delirium incidence (OR: 1.45; 95% CI: 1.12–1.89; p = 0.008) (Table 3).
The observed association between anticholinergic burden and delirium can be interpreted through the cholinergic hypothesis. In the oldest-old, the basal forebrain-cortex-hippocampus pathway, essential for attention and arousal, is particularly vulnerable. Systemic infection may further increase blood-brain barrier (BBB) permeability, allowing peripheral pro-inflammatory cytokines, such as IL-6 and TNF-α, to enter the central nervous system. This cascade leads to central neuroinflammation, which, when combined with the pharmacological blockade of muscarinic receptors, may synergistically impair cholinergic transmission and precipitate cognitive failure.
In this context, the results of our study suggest that the care burden in septic elderly patients is multidimensional [15]. The reduction in anticholinergic burden observed at discharge indicates that a focused geriatric approach, addressing not only the acute infection but also early rehabilitation and clinical stabilization, appears to be effective [16–18].
Our findings highlight that sarcopenia and pre-existing cognitive impairment may act as a “fertile ground” upon which pharmacological toxicity (anticholinergic load) and acute infection can contribute to a confusional state, subsequently exacerbating the care demands placed on healthcare staff and caregivers. Therefore, medication reconciliation and the systematic deprescribing of inappropriate anticholinergic drugs emerge as priority interventions to potentially reduce intra-hospital complications and improve clinical outcomes in the oldest-old [19–23].
In conclusion, optimizing care for hospitalized elderly patients requires a comprehensive perspective that extends beyond organ-specific pathology. While our findings suggest a strong link between anticholinergic burden and multidimensional vulnerability, the retrospective nature of this study precludes the establishment of a direct causal relationship. Therefore, this work should be considered a pilot study; we acknowledge that the small sample size (n = 60) represents an intrinsic limitation, and further prospective studies are necessary to collect larger cohorts, improve statistical power, and confirm the robustness of these observations.
However, a notable and distinctive feature of this study is that it represents the first work to concurrently analyze anticholinergic burden, sarcopenia, and delirium through the integration of Point-of-Care Ultrasound (POCUS). This methodology proved to be not only accurate but also simple, reproducible, cost-effective, and, most importantly, feasible at the bedside. The POCUS approach eliminates the need to transport frail patients to radiology departments, a process that is frequently unnecessary, distressing, and potentially harmful. The combined use of validated, streamlined tools (including the 4AT for delirium screening and the Beers Criteria for medication reconciliation) constitutes a practical clinical protocol readily applicable in routine geriatric care.
In summary, the information gathered at ward admission through these simplified scales and bedside muscle ultrasound offers valuable clinical insights for the geriatrician. In a context where frailty is increasingly prevalent, it is essential to recognize its two faces: cognitive-behavioral frailty, expressed through delirium, and physical frailty, manifested as sarcopenia. A specific focus on these two dimensions is vital for improving clinical outcomes, maintaining functional autonomy, reducing adverse events such as falls, and ultimately facilitating patient deinstitutionalization
This study was conducted in accordance with the Declaration of Helsinki. The protocol was submitted to the Institutional Ethics Committee and received approval (Reference ID: 2.116.999).
Written informed consent was obtained from all participants at the time of admission, covering the use of their pseudonymized data for clinical and research purposes. All data were anonymized and accessed only by the treating physician to ensure patient confidentiality.
Declaration of Helsinki STROBE Reporting GuidelineThis study adhered to the Helsinki Declaration. The Strengthening the Reporting of Observational studies in Epidemiology (STROBE) reporting guideline was followed.
The dataset of the study is available from the authors upon reasonable request.
The author declares that he has no conflicts of interest.
This research received no external funding.
The author would like to thank the clinical staff of the Geriatrics Operating Unit at San Luigi Gonzaga University Hospital for their support during the data collection phase.
1.
2.
3.

Copyright © Hapres Co., Ltd. Privacy Policy | Terms and Conditions